Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Tomita, Ryohei; Tomita, Jumpei; Suzuki, Daisuke; Yasuda, Kenichiro; Miyamoto, Yutaka
Hosha Kagaku, (48), p.1 - 15, 2023/09
Secondary Ion Mass Spectrometry (SIMS) is the method to detect secondary ions produced by the sputtering of primary ions. SIMS is one of effective method to measure isotopic composition of particles containing nuclear material in environmental sample for safeguards. We are a group member of the International Atomic Energy Agency (IAEA)'s network of analytical laboratories and have developed analytical techniques using SIMS and other mass spectrometers for nuclear safeguards. We will introduce the principle of SIMS and analytical techniques developed by our group to measure isotopic composition of uranium particles which having a particle diameter of micron order in environmental sample for safeguards.
Kabumoto, Hiroshi; Nakagawa, Sohei; Matsuda, Makoto
JAEA-Conf 2022-002, 146 Pages, 2023/03
"The 34th Meeting for Tandem Accelerators and their Associated Technologies" was held on July 21-22, 2022 organized by Nuclear Science Research Institute of the Japan Atomic Energy Agency. This meeting was held only on-line for preventing the spread of COVID-19 infection. The purpose of this meeting is contribution of development for related technology and of management of facilities through exchange of information among the researchers and engineers using and operating electrostatics accelerator facilities like tandem accelerators. There were 25 presentations which contains current status report of facility, technical development of accelerator, research of application. The total number of participants was a hundred, from 26 universities, research organizations and industries. This meeting consisted of only oral session, a poster session was not carried out because of on-line meeting. This proceeding compiles the contents of report papers in the meeting.
 Zr determination with isotope dilution ICP-MS
Zr determination with isotope dilution ICP-MSAsai, Shiho; Hanzawa, Yukiko; Konda, Miki; Suzuki, Daisuke; Magara, Masaaki; Kimura, Takaumi; Ishihara, Ryo*; Saito, Kyoichi*; Yamada, Shinsuke*; Hirota, Hideyuki*
Talanta, 185, p.98 - 105, 2018/08
Times Cited Count:9 Percentile:30.92(Chemistry, Analytical)Estimating the risks associated with radiation from long-lived fission products (LLFP) in radioactive waste is essential to ensure the long-term safety of potential disposal sites. In this study, the amount of  Zr, a LLFP, was determined by ICP-MS after separating Zr from a spent nuclear fuel solution using a microvolume anion-exchange cartridge (TEDA cartridge). The TEDA cartridge achieved highly selective separation of Zr regardless of its small bed volume of 0.08 cm
Zr, a LLFP, was determined by ICP-MS after separating Zr from a spent nuclear fuel solution using a microvolume anion-exchange cartridge (TEDA cartridge). The TEDA cartridge achieved highly selective separation of Zr regardless of its small bed volume of 0.08 cm . The time taken to complete the Zr separation was 1.2 min with a flow rate of 1.5 mL/min, which was 10 times faster than that for a conventional anion-exchange resin column. Almost all the other elements were removed, leading to accurate measurement of
. The time taken to complete the Zr separation was 1.2 min with a flow rate of 1.5 mL/min, which was 10 times faster than that for a conventional anion-exchange resin column. Almost all the other elements were removed, leading to accurate measurement of  Zr. The result connects experimental value to theoretical prediction provided by ORIGEN2, which requires verification. With the measured value, we demonstrated that the theoretical value is reliable enough to estimate radiation risks.
Zr. The result connects experimental value to theoretical prediction provided by ORIGEN2, which requires verification. With the measured value, we demonstrated that the theoretical value is reliable enough to estimate radiation risks.
 by Knudsen effusion mass spectrometry
by Knudsen effusion mass spectrometryNakajima, Kunihisa; Takai, Toshihide; Furukawa, Tomohiro; Osaka, Masahiko
Journal of Nuclear Materials, 491, p.183 - 189, 2017/08
Times Cited Count:12 Percentile:70.49(Materials Science, Multidisciplinary)One of the main chemical forms of cesium in the gas phase during severe accidents of light water reactor is expected to be cesium metaborate, CsBO , by thermodynamic equilibrium calculation considering reaction with boron. But accuracy of the thermodynamic data of gaseous metaborate, CsBO
, by thermodynamic equilibrium calculation considering reaction with boron. But accuracy of the thermodynamic data of gaseous metaborate, CsBO (g), has been judged as poor quality. Thus, Knudsen effusion mass spectrometric measurement of CsBO
(g), has been judged as poor quality. Thus, Knudsen effusion mass spectrometric measurement of CsBO was carried out to obtain reliable thermodynamic data. The evaluated values of standard enthalpy of formation of CsBO
was carried out to obtain reliable thermodynamic data. The evaluated values of standard enthalpy of formation of CsBO (g),
(g), 
 H
H
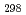 (CsBO
(CsBO ,g), by the 2nd and 3rd law treatments are -700.7
,g), by the 2nd and 3rd law treatments are -700.7 10.7 kJ/mol and -697.0
10.7 kJ/mol and -697.0 10.6 kJ/mol, respectively, and agree with each other within the errors, which suggests our data are reliable. Further, it was found that the existing data of the Gibbs energy function and the standard enthalpy of formation agreed well with the values evaluated in this study, which indicates the existing thermodynamic data are also reliable.
10.6 kJ/mol, respectively, and agree with each other within the errors, which suggests our data are reliable. Further, it was found that the existing data of the Gibbs energy function and the standard enthalpy of formation agreed well with the values evaluated in this study, which indicates the existing thermodynamic data are also reliable.
Konda, Miki; Asai, Shiho; Hanzawa, Yukiko; Magara, Masaaki
JAEA-Technology 2015-054, 22 Pages, 2016/03
Isotope dilution mass spectrometry (IDMS) with ICP-MS is reliable method for determination of Zr-93, which is one of the long-lived fission products found in spent nuclear fuel and high-level radioactive wastes. In order to use an isotope standard solution of zirconium as the spike for IDMS, dissolving a commercially available solid isotope standard is indispensable. Prior to the dissolution of the Zr-91 isotope standard, solubility of metal zirconium in a mixture of HNO and HF was evaluated using zirconium metal chips. Then, 2 mg of the Zr-91 isotope standard was dissolved with 0.2 mL of 1 M HNO
and HF was evaluated using zirconium metal chips. Then, 2 mg of the Zr-91 isotope standard was dissolved with 0.2 mL of 1 M HNO -3 v/v% HF mixed solution, followed by adjusting the concentration of Zr-91 to approximately 1,000
-3 v/v% HF mixed solution, followed by adjusting the concentration of Zr-91 to approximately 1,000  g/g. IDMS, in which a natural isotopic abundance standard solution of zirconium was used as the spike, was employed for the determination of the concentration of Zr-91 in the prepared Zr-91 isotope standard solution. The concentration of Zr-91 in the prepared Zr-91 isotope standard solution was (9.6
g/g. IDMS, in which a natural isotopic abundance standard solution of zirconium was used as the spike, was employed for the determination of the concentration of Zr-91 in the prepared Zr-91 isotope standard solution. The concentration of Zr-91 in the prepared Zr-91 isotope standard solution was (9.6 1.0)
1.0)  10
10
 g/g, which is in good agreement with the predicted concentration. This indicates that the Zr-91 metal isotope standard was completely dissolved with sufficient chemical stability. Additionally, no impurities were detected in the prepared Zr-91 isotope standard solution. These positive results denote that the Zr-91 isotope standard solution with the preferable quality for IDMS of Zr-93 can be obtained by the proposed dissolution procedures.
g/g, which is in good agreement with the predicted concentration. This indicates that the Zr-91 metal isotope standard was completely dissolved with sufficient chemical stability. Additionally, no impurities were detected in the prepared Zr-91 isotope standard solution. These positive results denote that the Zr-91 isotope standard solution with the preferable quality for IDMS of Zr-93 can be obtained by the proposed dissolution procedures.
 I in environmental samples by AMS and NAA using an anion exchange resin disk
I in environmental samples by AMS and NAA using an anion exchange resin diskSuzuki, Takashi; Bamba, Shigeru*; Kitamura, Toshikatsu; Kabuto, Shoji*; Isogai, Keisuke*; Amano, Hikaru
Nuclear Instruments and Methods in Physics Research B, 259(1), p.370 - 373, 2007/06
Times Cited Count:15 Percentile:68.94(Instruments & Instrumentation)no abstracts in English
Yamamoto, Hiroyuki; Saito, Takeru; Asaoka, Hidehito
Journal of Trace and Microprobe Techniques, 19(4), p.571 - 579, 2001/11
Times Cited Count:4 Percentile:14.78(Chemistry, Analytical)no abstracts in English
Yamamoto, Hiroyuki; Saito, Takeru; Asaoka, Hidehito
Applied Surface Science, 178(1-4), p.127 - 133, 2001/07
Times Cited Count:5 Percentile:31.87(Chemistry, Physical)no abstracts in English
Esaka, Fumitaka; Zheng, W.*; Watanabe, Kazuo; Magara, Masaaki; Hanzawa, Yukiko; Usuda, Shigekazu; Adachi, Takeo
Advances in Mass Spectrometry, 15, p.973 - 974, 2001/00
no abstracts in English
Kato, Kaneharu; Ito, Mitsuo; Watanabe, Kazuo
Fresenius Journal of Analytical Chemistry, 366(1), p.55 - 58, 2000/01
no abstracts in English
; Mita, Naoaki; Nishino, Yasuharu; Amano, Hidetoshi
JAERI-Conf 99-009, p.103 - 111, 1999/09
no abstracts in English
 C=C
C=C
Hayakawa, Shigeo*; ; Arakawa, Kazuo; Morishita, Norio
Journal of Chemical Physics, 110(6-8), p.2745 - 2748, 1999/02
Times Cited Count:22 Percentile:56.50(Chemistry, Physical)no abstracts in English
Yamamoto, Hiroyuki; Baba, Yuji
Applied Physics Letters, 72(19), p.2406 - 2408, 1998/05
Times Cited Count:9 Percentile:42.66(Physics, Applied)no abstracts in English
 H
H isomers by charge inversion mass spectrometry
isomers by charge inversion mass spectrometryHayakawa, Shigeo*; ; Arakawa, Kazuo; Morishita, Norio
Int. J. Mass Spectrom. Ion Process, 171(1-3), p.209 - 214, 1997/00
no abstracts in English
Sekiguchi, Tetsuhiro; Selriguehi, T.; Sekiguchi, Tetsuhiro; Sekiguchi, Hiromi*; Obi, Kinichi*; Tanaka, Kenichiro*
Journal de Physique, IV, 7(C2), p.505 - 506, 1997/00
no abstracts in English
Sekiguchi, Hiromi*; Sekiguchi, Tetsuhiro
Surface Science, 390(1-3), p.214 - 218, 1997/00
Times Cited Count:9 Percentile:52.41(Chemistry, Physical)no abstracts in English
Sekiguchi, Tetsuhiro; Sekiguchi, Hiromi*
Atomic Collision Research in Japan, 0(22), p.89 - 90, 1996/00
no abstracts in English
 H
H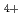 isomeric ions by charge inversion mass spectrometry using an alkali metal target
isomeric ions by charge inversion mass spectrometry using an alkali metal target; ; Arakawa, Kazuo; Morishita, Norio;
Int. J. Mass Spectrom. Ion Process, 151, p.89 - 95, 1995/00
no abstracts in English